ECCO 2024 is coming soon!

Social distancing time seems far away, and we already enjoyed meeting face to face last year. Let’s continue in 2024!
For the first time, the congress of the European Crohn’s and Colitis organization (ECCO) congress will take place in Stockholm, from February 21st to 24th.
BÜHLMANN Highlights
New CALEX® Cap Stool Collection Set Tutorial Video
The CALEX® Cap Collection Set offers a convenient solution to collect stool samples. The CALEX® Cap stool preparation device enables the measurement of both fecal calprotectin and pancreatic elastase from a single extraction tube.
IBDoc® Calprotectin Home Test Tutorial Video
IBDoc® is a user-friendly home testing device designed for the quantitative assessment of fecal calprotectin levels at home. With simple steps, it enables the self-testing of calprotectin levels in IBD patients from the comfort of their own homes. We are getting ready to release an update aiming to increase the usability and stability of our at home calprotectin test in order to further help patients and healthcare providers. Come to our booth to check it out!
IBDoc® Success Story
Professor Patrick van Rheenen, a distinguished pediatrician based at the University Medical Center Groningen (UMCG) in the Netherlands, shared insights into the seamless incorporation of the IBDoc® fecal calprotectin home test into a hospital’s remote monitoring system. This achievement was made possible through the utilization of the standard HL7 communication pathway.
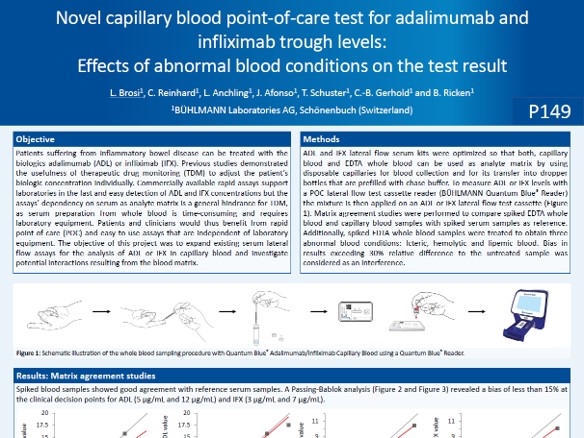
BÜHLMANN Quantum Blue® Capillary Blood Assays for Therapeutic Drug Monitoring
We presented at the ECCO 2023 a poster comparing Therapeutic Drug levels between serum, EDTA whole blood and capillary blood samples. It enables healthcare professionals to perform anti-TNFα monitoring by measuring biologics levels, using capillary blood or EDTA whole blood samples. These assays offer a remarkable time to results of just 15 minutes, eliminating the need for additional laboratory equipment.
BÜHLMANN Posters
We are pleased to present 2 posters this year at ECCO 2024!

BÜHLMANN Calprotectin and therapeutic drug monitoring (TDM) Assays
Inflammatory Bowel Disease (IBD) includes Crohn’s Disease (CD) and Ulcerative Colitis (UC), two currently incurable serious chronic diseases of the intestinal tract. IBD symptoms strongly resemble those of non-inflammatory diseases such as irritable bowel syndrome (IBS). Fecal calprotectin has proven itself as an excellent surrogate marker in inflammatory bowel disease diagnosis and helps to reliably select patients for further invasive diagnostic procedures. Further, calprotectin in stool can also be used for Crohn or Ulcerative Colitis monitoring in diagnosed patients since it correlates closely with mucosal healing. High calprotectin values are indicative for active inflammation in the gut, while moderate fecal calprotectin values can represent mild organic disease such as Crohn or Ulcerative Colitis. On the other hand, a low calprotectin value can indicate a remissive disease. In addition to fecal calprotectin, the monitoring of biologic drug trough levels (therapeutic drug monitoring or TDM) as part of the therapeutic follow-up in IBD patients is becoming standard care. BÜHLMANN offers the broadest product portfolio supporting decision making in the inflammatory bowel disease treatment management.
The product portfolio includes the FDA cleared BÜHLMANN fCAL® turbo for high throughput testing. The immuno-turbidimetric fecal calprotectin assay BÜHLMANN fCAL® turbo provides flexible and random-access applications on all major clinical chemistry platforms. The assay allows short turnaround time from fecal sample to the reportable result. The combination of the test with the CALEX® Cap device or the fecal collection kit CALEX® Cap Collection Set strongly simplifies the cumbersome procedure for stool pre-analytics and is adapted to be applied on clinical chemistry analyzers. BÜHLMANN also offers the FDA cleared market leader Fecal calprotectin ELISA (BÜHLMANN fCAL® ELISA) and the Quantum Blue® calprotectin rapid tests. Moreover, the portfolio includes IBDoc®, a highly successful inflammatory bowel disease home test for patients using an app that transforms smartphones into a calprotectin test reading device.

The BÜHLMANN Quantum Blue® rapid test combines the ease and speed of lateral flow technology, with full quantification by means of a small reading device. The Quantum Blue® Reader provides a quantitative value within minutes. The BÜHLMANN product line offers two different fecal calprotectin test ranges suitable for inflammatory bowel disease diagnosis and therapy follow-up of Crohn or Ulcerative Colitis patients. Besides calprotectin assays, BÜHLMANN offers the possibility for therapeutic drug monitoring for biologics in IBD on the Quantum Blue® Reader.

IBDoc® is the first in vitro diagnostic home testing device measuring the inflammatory marker fecal calprotectin at home. The IBDoc® mobile app turns your smartphone into a test cassette reader using state of the art image processing. Stool preparation is performed using the CALEX® Valve that is characterized by its simple and convenient handling of stool samples. The secure data connection between the IBDoc® mobile app and the IBDoc® Portal allows Health Care Professionals to directly monitor Crohn’s or Ulcerative Colitis’ patient results. The IBDoc® Portal also includes an application programming interface (API) that directly transfers test results into electronic patient records (EHR) or laboratory information systems (LIMS) via a common web standard or HL7 based interface.

The CALEX® Cap Collection Set further reduces the laboratory task of stool preparation procedure for fecal calprotectin and pancreatic elastase testing with BÜHLMANN assays. The CALEX® Cap Collection Set offers efficient, convenient, and hygienic direct stool collection.
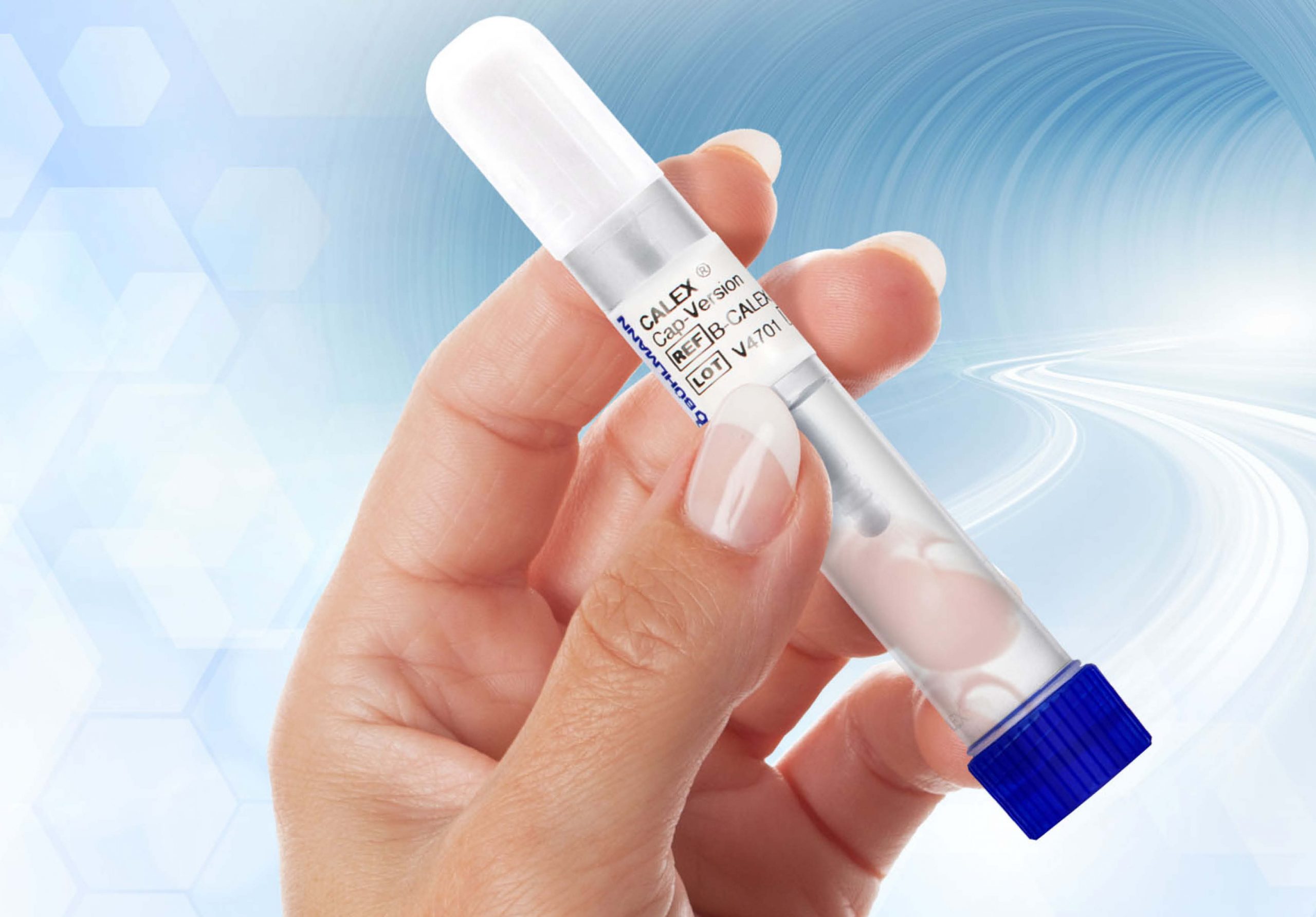
The BÜHLMANN CALEX® Cap device simple design and buffer offer the user high safety as well as high stability of stool samples. With the device, stool collection and extraction in the laboratory environment is efficient, convenient, and hygienic. Furthermore, the CALEX® Cap is designed to be fully compatible with total laboratory automation (TLA) solutions.

The new assay BÜHLMANN fPELA® turbo and the well-established BÜHLMANN fCAL® turbo together with the CALEX® Cap have revolutionized fecal testing. Both turbidimetric immunoassays can be easily applied for a wide choice of clinical chemistry analysers, providing TLA with fast turn-around-time (TAT) and improve laboratory workflow. The combined use of these products makes reliable results possible for an extended measuring range.

BÜHLMANN launched its first assay for fecal calprotectin testing more than 15 years ago and is today considered as the gold standard with the BÜHLMANN fCAL® ELISA, a trusted tool applied in numerous large clinical trials. Two protocols are available with the BÜHLMANN fCAL® ELISA to cover different calprotectin ranges for Crohn and Ulcerative Colitis diagnosis and monitoring.